SAFETY IN THE USE OF LPG
All our flame heat treatment machines or equipment use LPG as fuel.
Heat treatment is the working technique for controlling or eliminating weeds, fungal diseases, harmful insects and sterilising the surface of the treated soil by means of HEAT generated by a flame
See: Heat treatment with flame
From an ecological point of view flame heat treatment is a practice characterised by a completely negligible environmental impact..
Since LPG burning only forms water vapour and carbon dioxide, the flame is transparent and free of any smoke release phenomenon, any type of flooring or covering, which is not subject to combustion, can therefore be directly invested with the flame without altering its colour.
The heating of the surface layers of the soil, caused by the rapid passage of the burners, brings the surface itself to a temperature that generally does not exceed 80-90°C, a temperature that can easily be found even in the hottest hours of the summer season.
There is therefore negligible damage to the soil’s microflora and micro-organisms; there are no risks to operators and no toxic residues are released into the environment.
Another obtainable result, from the point of view of environmental hygiene, is that which can be achieved by slowing down the operation. Slowing down the speed at which the treatment is applied brings the surface of the ground, pavement or concrete floors to slightly higher temperatures (90–100°C); this deprives them of pathogenic germs, allowing an effective sterilisation operation
With the application of this technology, the costs involved can be determined immediately, since it does not give rise to environmental pollution and any additional costs following the treatment carried out and due to the disposal of any toxic residues are therefore nil.
Gas consumption is closely linked to the type of equipment in use and the result required by the treatment, the environmental conditions and any plants treated.
WHAT IS LPG
The acronym G.P.L. is used to identify “liquefied petroleum gases”, hydrocarbons and their mixtures which can be easily liquefied under moderate pressures , because of their constant criticality and low vapour pressure.
It is a commercial product that is obtained:
- from the processing of crude oil
- from natural gas deposits
Under the name of G.P.L. are defined:
- commercial propane
- the commercial butane
- commercial mixtures of propane and butane
LPG is:
- an environmentally friendly energy source: low pollutant emissions into the atmosphere;
- clean (pure): the sulphur content of G.P.L. is almost absent;
- colourless: artificially coloured/denatured for fiscal reasons;
- Odourless: it is odorized so that its presence can be detected in case of leaks;
- non-poisonous: it is not poisonous even at high concentrations, but an individual can succumb to asphyxia if locked in a room with a high percentage of G.P.L.;
- non-corrosive: not corrosive as it is not present in the G.P.L. sulfur in active form (hydrogen sulfide);
- high calorific value
- Easily usable, storable and transportable
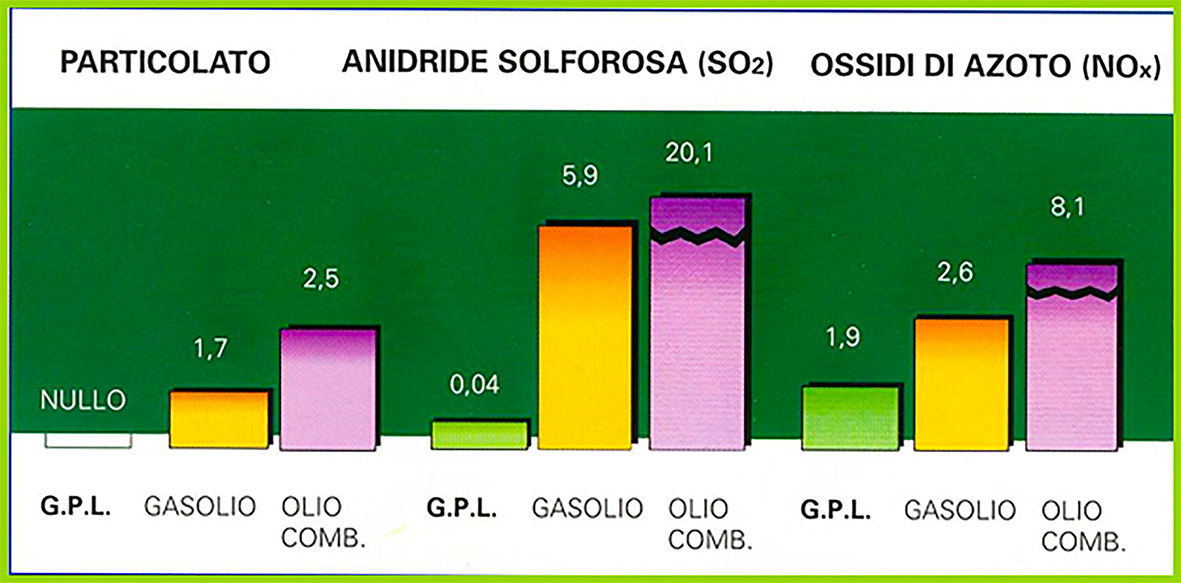
AIR POLLUTION
The combustion of G.P.L., at the same burned energy, compared to that of diesel and fluid fuel oil, in heating highlights the absence of particulates, negligible presence of SO2 and low emission of NOX.
COMBUSTION
C The term “combustion” refers to a chemical combination of oxidation between a FUEL substance and a COMBURENT (oxygen) with development of thermal energy, this combination occurs very quickly.
To ignite combustion it is necessary:
- to produce a homogeneous mixture of combustible and oxidising gas in such proportions that it is flammable;
- Raise the temperature at any point in the mixture to a value equal to or greater than the ignition temperature.
To ensure constant combustion, it is necessary:
- Evacuate combustion fumes;
- Ensure continuity of fuel gas and combustion air supply to the burner.
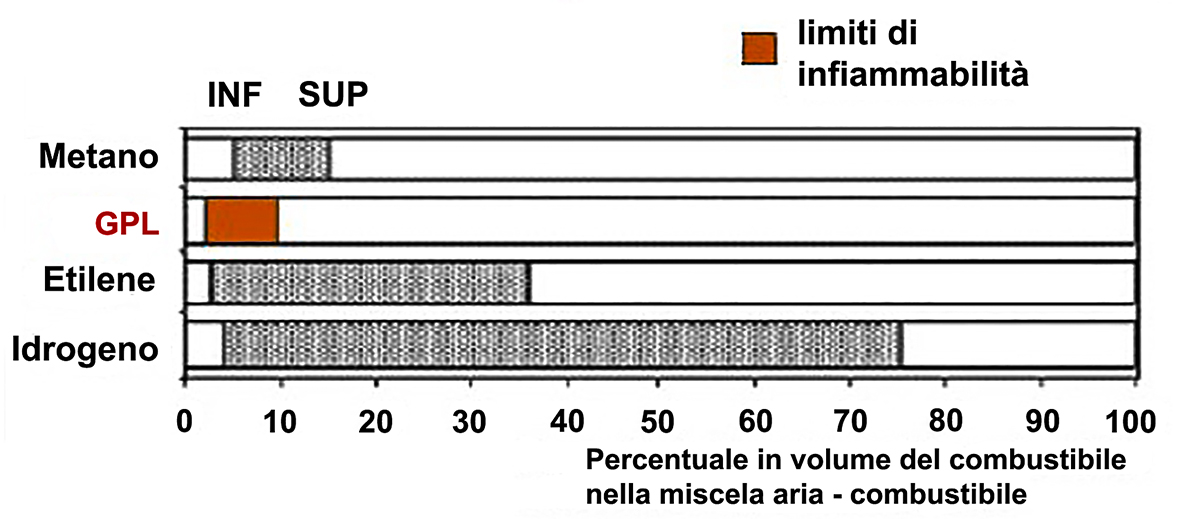
FLAMMABILITY LIMITS
Fuel-air mixtures are only flammable within a range of concentrations defined by the lower limit Li and the upper limit Ls.
The tighter these limits are, the safer the fuel is since its flammability conditions are non-existent.
For example, with air in standard conditions: 25°C and atmospheric pressure (1.013 bar) the percentage by volume concentration of LPG necessary to give rise to the reaction is between 2% and 9% of the total volume of the mixture ARIA – LPG. These values are called the flammability limits (lower and upper) of the fuel.
Comparison of the flammability limit of LPG with other fuels
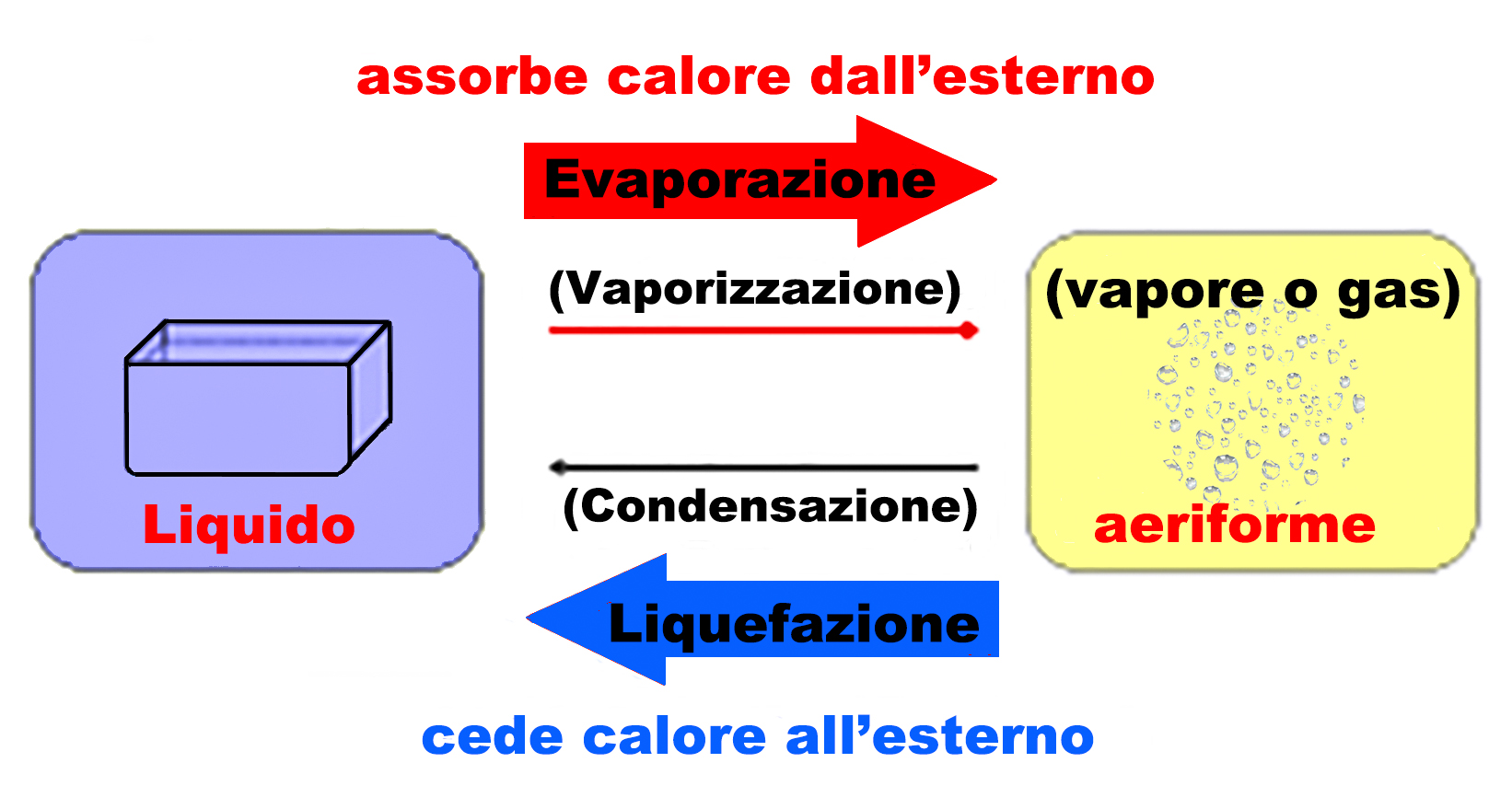
PHYSICAL TRANSFORMATIONS
One of the characteristics of the three states in which we can find matter around us is determined by the interaction between the molecules that constitute it.
If the interaction is strong the matter will tend towards solid, if weak towards liquid. If the molecules are even far enough apart from each other, the matter will be aeriform (in common parlance vapour or gas).
This is why solids generally have a higher density than liquids and gases. Hence the ability of liquids and aeriformes to take on the shape of the container they are in.
Solids and liquids also retain their volume and are incompressible , at not particularly high pressures.
Aeriforms, on the other hand, can be compressed or expanded by varying the pressure to which they are subjected.
State transitions or phase changes take on different names depending on the starting state and the target state.
For each substance, these transitions occur at different pressures and temperatures, which are intrinsic to the substance itself.
Matter possesses the ability to transform its physical state when subjected to changes in temperature or pressure or both of these quantities.

LPG TANKS
The container, usually horizontal or vertical cylindrical in shape, is about 80% filled with LPG in the liquid phase, while the remaining 20% volume contains LPG vapour.
The equilibrium between the two phases (vapour pressure) is dynamic, in the sense that at equilibrium pressure (also known as saturated vapour pressure) the mass of the liquid and vapour do not change, even though liquid molecules are continually turning into vapour (vaporisation) and vapour molecules are continually turning into liquid (liquefaction).
To break the equilibrium, steam must be removed. For example, if we open the cylinder tap and use LPG, vapour is removed. This is immediately reproduced by the liquid mass, which then evaporates during use by cooling.
After closing the valve, the equilibrium between liquid and aeriform propane is restored, with pressure values not far from the initial ones. At ordinary temperatures, this results in pressures close to 10 bar (ten times, approximately, the atmospheric pressure of 1 bar). Thus, given the considerable difference between the density of the liquid and the gas, it is possible to obtain approximately 270 litres of vapour per litre of liquid propane.
All our machines or equipment are sold with automatically triggered safety valves that block the release of LPG from the cylinder in the event of excess demand for it from the burner supply system (e.g. in the event of a tear or rupture of a tube connecting the LPG cylinder(s) to the machine)
In the event of an abnormal LPG leak from the machine supply system, the degree of danger linked to the flammability of the fuel lies in the quantity that is outside the cylinder(s) at that moment.
In all our machines and equipment, this amount of LPG is proportional to the machine/equipment model, but for each of them it reduces the time for eventual combustion within the minute
TUT ALL OUR MACHINERY AND EQUIPMENT ARE FUelled WITH LPG IN A GASEOUS PHASE

